Nortech can design your innovative technology specifically for regulated medical device applications. This includes risk mitigation strategies, software design, program management and supporting documentation for regulatory submissions. Using our proven process, your project can move much faster to commercialization.
Nortech can design your innovative technology specifically for regulated medical device applications. This includes risk mitigation strategies, software design, program management and supporting documentation for regulatory submissions. Using our proven process, your project can move much faster to commercialization.

Nortech Systems is an ISO 13485-certified and 14971-compliant, multi-disciplinary engineering and manufacturing firm. We deliver innovative design solutions and comprehensive product development services to the medical device industry. Nortech is a full-service device and lifecycle management solutions provider.

Start with the end in mind and consider assembly, testing, and serviceability requirements. Partner with an EMS provider that can act as a single point of contact for all engineering and production needs and maintains both a robust and flexible quality system.

Provide structure for project delivery through five coordinated phases to keep everything on track. We will track and meet your project needs by customizing our process to your budget and resources.

Move from tactical order placement to strategic demand planning for better inventory availability, cost planning, and revenue actualization.

We understand the importance of post-market support to the safety and reliability of your products. We offer value-added services, including automated replenishment, repair, refurbishment, recycling, product certification and compliance testing, and warranty support.
Medical-grade solutions require the highest level of design and support. When medical devices are designed with Design for Manufacturability [DFM] in mind, you can reduce prototype iterations; streamline the production process, making it more efficient and cost-effective; and ensure that efficient testing and servicing options are not an afterthought. In addition, medical devices must meet stringent safety and quality standards, and a manufacturing company must have the necessary expertise to produce reliable, high-quality products.
Startups lacking a formal or experienced quality system can rely on our ISO 13485 certified product development quality system to serve as their own. For companies with an established quality system, the appropriate documentation will be created and transferred to their system according to their protocols.
Unlike many other vendors, Nortech Systems can design your innovative technology specifically for regulated medical device applications, including risk mitigation strategies, human factors analysis, and supporting documentation for regulatory body submissions. We offer a complete suite of services, from concept development and feasibility studies to product design and development, prototyping, and manufacturing support.

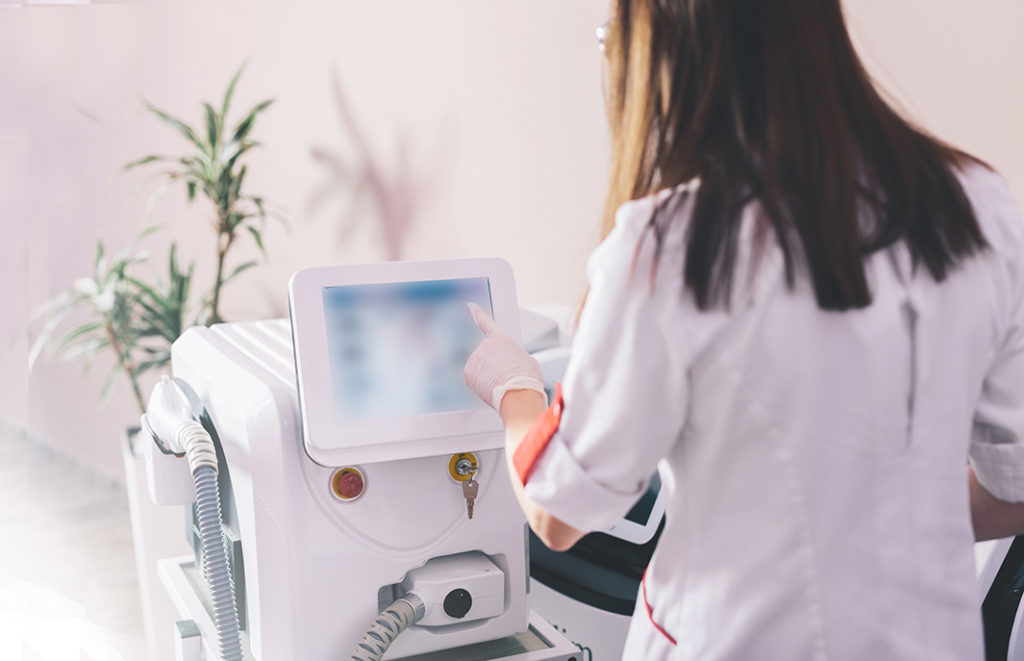
Commercialization of medical devices can be a long and challenging process, particularly for products that are produced in low volumes or have a high mix of components. Our Mankato and Bemidji, MN locations are tailor-made for these scenarios.
An excellent medical device contract manufacturer will also be able to quickly ramp up production to meet the market’s demands while maintaining a high level of quality control. As volumes reach a higher threshold, Nortech has facilities in China and Mexico available to accommodate those production needs as well.
By working with a medical device contract manufacturer that can offer scalable solutions, you can get your products to market faster and with less risk. In addition, we can provide support throughout the product development process, from design and prototyping to manufacturing, packaging, testing, shipment, and post-market support.
Using our advanced technology and designs, your project can move swiftly to commercialization compared to a project that starts from scratch. Our vendor partners work with us directly so we can support all aspects of your rollout, including supply chain management. Let us reduce the time and money you spend managing vendors and subcontractors by becoming your one point of contact for all.
Are you ready to get your medical-grade solution to market faster and work with a company that supports the entire lifecycle of your product?
Medical device manufacturing is a complex and regulated process. In order to bring your medical-grade solution to market, you need to work with a partner who understands the challenges and can support you through the entire lifecycle of your product.
Eliminate unnecessary prototype iterations, mitigate the risks of scaling and transitioning your design to production, and efficiently test, repair, and service your medical devices and components at all product lifecycle stages.
Work with an ISO 13485 certified manufacturer and know that adequate testing is performed throughout the product development cycle. Board level testing reduces scrap in manufacturing and ensures consistently low DPPM levels resulting in medical devices of the highest quality and safety for patients.
Whether your design is still just a great idea, or you are ready to hand over your specifications and have contract manufacturing services take over, we work every day to earn your trust and deliver value back to you through solid relationships, expert design, and the right people.
Nortech Systems offers a full line of electronic manufacturing services, from design and development to commercialization and post-market support. We also offer a wide range of value-added services, such as Program Management and Quality Management, to help you navigate the product development landscape and ensure that your product meets your requirements.
We aim to make it easy for you to help bring innovative, life-changing products to market quickly and safely.
Post-market support for your medical-grade solutions should start before your specifications transition to production. A skillful contract manufacturing partner will take the time to consider the lifespan and service life expectations of your medical device and components to ensure they allow for various installation scenarios and field maintenance access.
As a medical device contract manufacturer, we understand the importance of post-market support. We offer testing design and implementation services to ensure your devices perform as expected. We also offer flexible shipment options, including automated warehouse replenishment. In addition, we provide repair, refurbishment, and recycling services to keep your devices in working order. We are committed to providing our customers with the highest level of service and support. Our experience and expertise can provide ongoing design, manufacturing, and post-market service options that offer value and peace of mind.
We design, develop and manufacture Class I, II, and III medical devices in numerous and various medical specialties, including but not limited to cardiology, orthopedic and surgical systems, point-of-care diagnostic instruments, and therapeutic devices (including stimulators). Nortech’s team has expertise in:
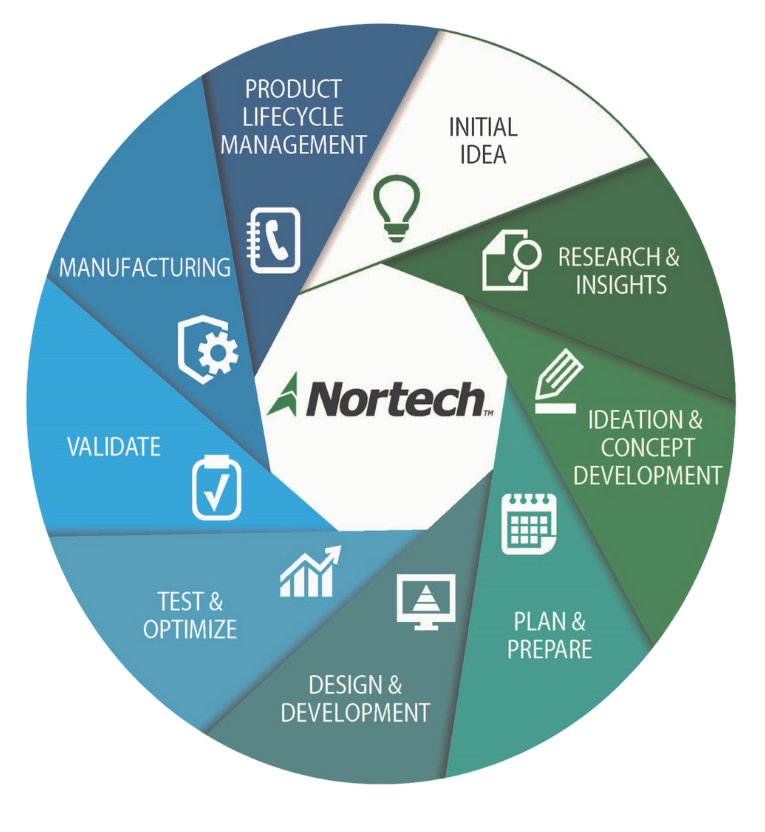
Nortech has more than 20 years of medical device development experience. We understand the U.S. FDA and international medical device approval processes and have a track record of successful approvals. Projects range from turnkey commercialization of technologies to task-based prototyping, post product service & support, and sustaining engineering.
Our extensive experience in medical device manufacturing and your willingness to engage in a joint DFM process can be leveraged to beat your competitors to market. Take advantage of our ability to identify and remedy design gaps that eliminate test iterations, ensure a smooth production transition, and keep your medical devices fully functioning and servicing your customer’s critical needs well into the future.
Whether you are still concepting or have a fully functional design, we are ready to engage with you in all phases of the product lifecycle. As always, our Lean 6 Sigma continuous improvement process and dedicated quality teams are always aimed at how to deliver more back to you through our certifications and best practices.
ISO 9001 Certified
ISO 13485 Certified
ISO 14971 Compliant
IEC 60601 Compliant (Electronic Medical Devices)
IEC 62304 Compliant (Medical Device Software)
IEC 62366 Compliant (Human Factors)
FDA Registered Facilities