Home / Table of Contents In industries where precision, performance and reliability are non negotiable, fiber connectivity is...
Read More »Our engineers use their knowledge of product design, biomedicine, electronics, mechanics, and control systems to design, develop, and manufacture Class I, II, and III medical devices. We have a development team exclusively focused on medical products, experienced in clinical applications of capital and disposable products, including cardiology systems, orthopedic equipment, surgical devices, point-of-care diagnostic instruments, and therapeutic devices.
Our engineers use their knowledge of product design, biomedicine, electronics, mechanics, and control systems to design, develop, and manufacture Class I, II, and III medical devices. We have a development team exclusively focused on medical products, experienced in clinical applications of capital and disposable products, including cardiology systems, orthopedic equipment, surgical devices, point-of-care diagnostic instruments, and therapeutic devices.

Some of our specific medical device design proficiencies include:
Our services also cover everything from finished products to subsystems and interconnects, and we provide qualification and acceptance test planning, tooling and fixturing, and mechanical and environmental ruggedization. Our team is skilled in architecture simplification, copper and optical hybrid technologies, and embedded diagnostics.
Perfectly tuned baluns can reduce electromagnetic interference and shield currents, ensuring an intrinsically safe patient environment. Reaching design consensus quickly, maximizing commercial success, and getting FDA approval with minimal effort will help you achieve fast results and superior patient care with confidence.
Having an electromechanical manufacturing services provider with expertise in complex balun design challenges is crucial for the success of a project. Early engagement with our team offering quick-prototyping to accelerate development, and on-site testing to ensure that all products meet the highest standards for quality, speed, and safety, allows you to maximize your potential revenue and client partnerships.
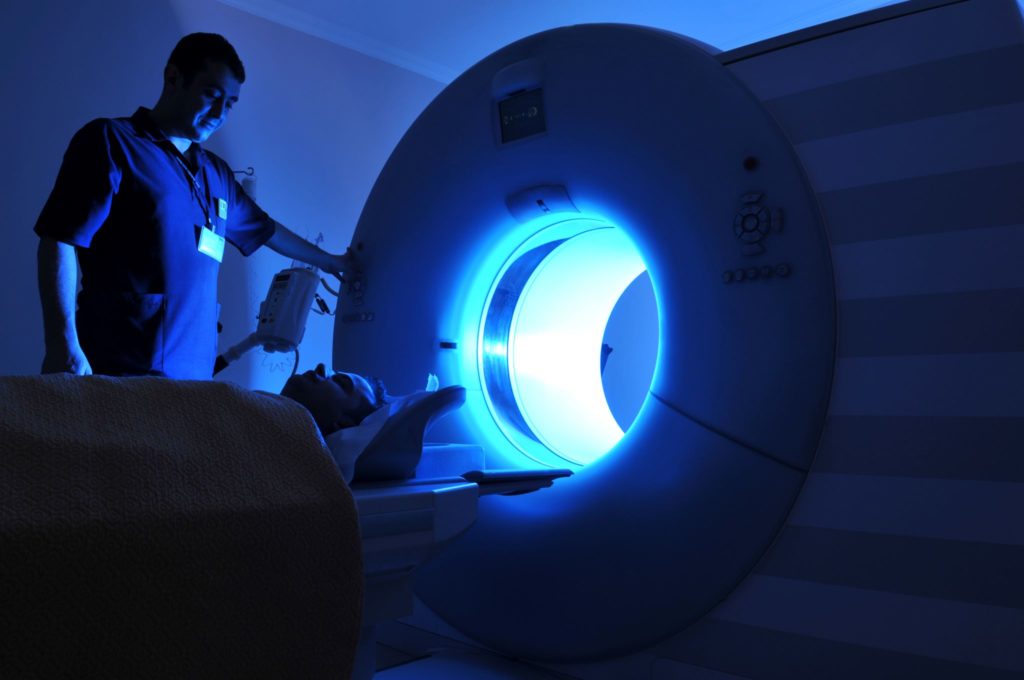
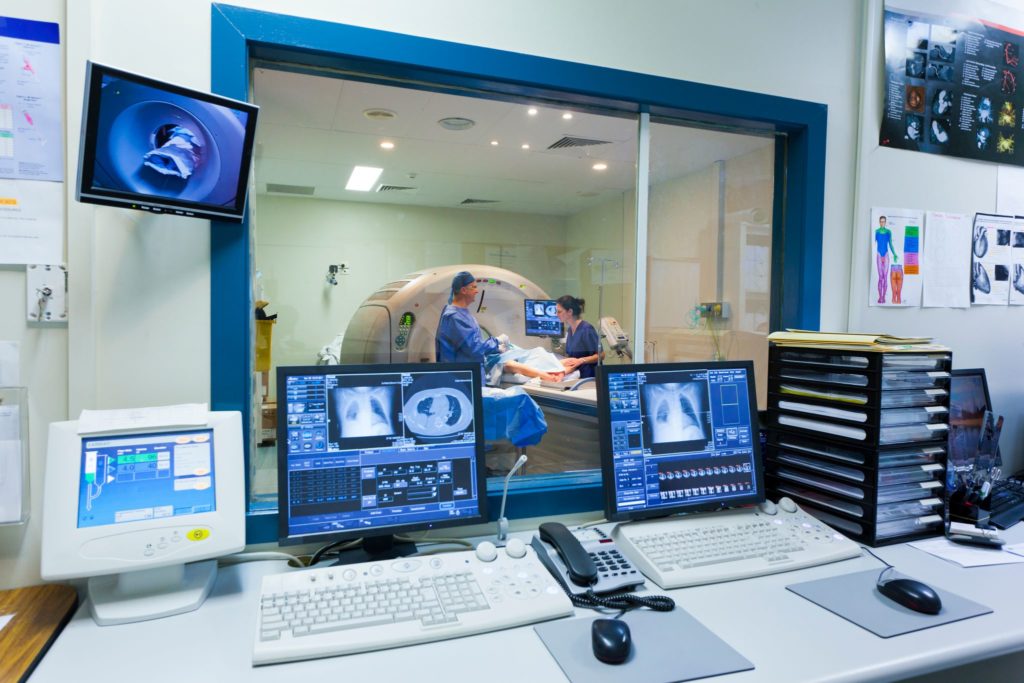
When developing custom software for medical devices, an engineering team experienced in this specialty area makes all the difference. Working with an engineering team that is familiar with and can expertly handle custom board design, power management, motor control, and safety-critical embedded software requirements, among many others, is essential in the development of medical devices for cardiology, orthopedic and surgical systems, point-of-care diagnostic instruments and therapeutic devices (including stimulators).
Through their extensive experience in this field, our team can anticipate potential problem areas and design solutions before they happen, resulting in a robust product with better accuracy, greater compliance to regulations, and a higher quality product that gets to market swiftly.


Accelerating the commercialization of medical device products requires a comprehensive approach, including deep expertise in design, development, testing, and project management to ensure that the product is functional, efficient, and meets the end user’s requirements.
Beginning with the end in mind, we leverage our extensive medical engineering and design expertise to simplify and accelerate the process while achieving time, cost, and quality goals. Our robust and flexible Quality Management System, with full component traceability, will also streamline the regulatory approval process. Frequent and complete communication from our customizable program management process, which works with any size company, will ensure that your production-ready product can launch into the market faster.
Our embedded team enabled fast prototyping, testing and manufacturing of the technology being developed. This provided our client with a rapid shift to manufacturing, providing the speed necessary for successful market delivery and allowing them to recognize the value of our team’s efforts.
Our team provided the solution to our client’s problem with managing multiple suppliers and design iterations. Nortech’s disciplined approach and flexible quality system created a clear and consistent path for device approval, eliminating unforeseen setbacks and minimizing gaps.
Through early engagement and leveraging our expertise, technology, and resources, we helped our client develop a detachable MRI table docking system. With our integrated team of engineers, daily progress updates, and data to streamline FDA approval, the technology designed and developed has been successfully implemented into healthcare facilities.
Are your projects being delayed because your engineering resources are over-allocated? Are inefficient design and testing leading to R&D cost overruns? Bring together your team with our in-house specialized resources to get everyone the answers they need on time and within budget.
Home / Table of Contents In industries where precision, performance and reliability are non negotiable, fiber connectivity is...
Read More »https://youtu.be/meo_Py5uqfg?si=kQGNtl8eN_fsu2Gg Addressing the Need for a New Solution In 2024, Nortech announced Expanded Beam Xtreme® as an expansion...
Read More »Wearable medical devices have evolved from simple fitness trackers to sophisticated diagnostic tools that monitor everything from blood...
Read More »High-Performance Flexible Circuit Flex Faraday Xtreme™ Military personnel operate in environments where equipment failure isn’t just inconvenient, it’s...
Read More »