Home / Table of Contents In industries where precision, performance and reliability are non negotiable, fiber connectivity is...
Read More »Nortech has been designing and manufacturing specialty wire, cable, and harness products for over four decades with the quality and dedication needed to create complex wire and cable projects. With this many years of design and production experience specifically for these types of applications, we know what it takes to design an interconnect to be reliable, meet your specifications, and offer incredible value to you and your customers.
Nortech has been designing and manufacturing specialty wire, cable, and harness products for over four decades with the quality and dedication needed to create complex wire and cable projects. With this many years of design and production experience specifically for these types of applications, we know what it takes to design an interconnect to be reliable, meet your specifications, and offer incredible value to you and your customers.

When designing electronic medical devices, connectors are often the last thing to be considered because the primary focus is typically on the functionality and performance of the device itself. Connectors are considered secondary to this and are often given little attention until the end of the design process. Additionally, the selection and integration of connectors can be complex and time-consuming, and designers may only want to spend resources on this once the device’s other aspects have been finalized.
However, connectors not compliant with regulatory standards and guidelines may not be approved for use, delaying or failing to bring the final product to market. Connectors not designed to safety standards can lead to malfunctions, recalls, and product liability issues. Weak connections can also cause malfunctions, increased maintenance and repair costs, and decreased device uptime.
At Nortech, we know that the interconnect solutions are a critical component of the device. We will apply our extensive knowledge base to selecting and integrating these interconnect solutions, thereby minimizing the negative impact on the safety, durability, compliance, reliability, and cost throughout the product’s lifecycle.

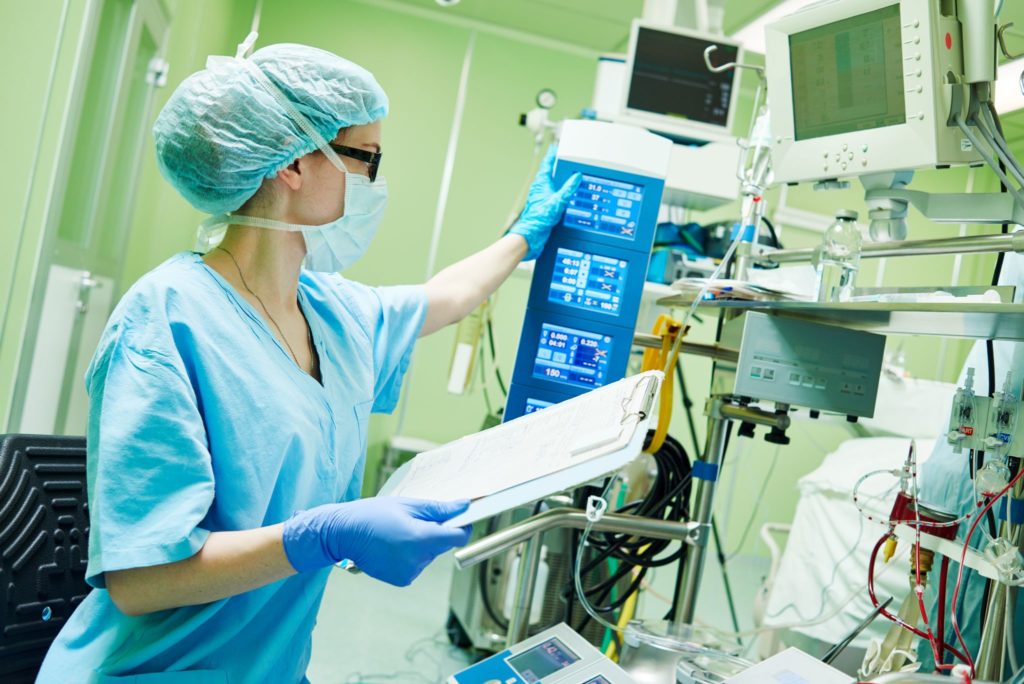
Medical devices used to diagnose and treat patients must be durable and reliable to function properly and transmit accurate information. These same devices are often at the center of critical situations and must also be able to withstand harsh environments and transmit that same data quickly, ensuring medical staff efficiency to keep the patient safe.
Designing specifically for medical interconnect applications is important because it ensures that the device will function correctly and safely in the specific environment in which it will be used. These design considerations can include factors such as extreme temperatures, electromagnetic interference, and radio frequency delivery, which must be carefully managed to prevent damage or malfunction of the device. By designing and manufacturing with these specific requirements in mind, Nortech can ensure that our medical interconnect solutions will meet your needs and be safe for use in medical environments.

Unauthorized access, tampering, and information leakage are some potential medical device cybersecurity threats that can be addressed through comprehensive interconnect solution selection and integration. However, it is important to note that security measures need to be designed not to impede the medical device’s primary function and availability to the patient.
Restricting access to certain connections by placing them inside devices where only authorized maintenance personnel can access them is one way to mitigate the threats. Using secure connections and protocols, including encrypted connections, such as HTTPS or SSH, to protect data transmitted over the connector is another threat mitigation approach.
To determine the most appropriate type of connector for a device, let our team conduct a cost/benefit analysis to weigh the costs of implementing different types of connectors and security measures against the potential benefits of those measures in terms of improved security, compliance with regulations, and lifecycle costs.
Home / Table of Contents In industries where precision, performance and reliability are non negotiable, fiber connectivity is...
Read More »https://youtu.be/meo_Py5uqfg?si=kQGNtl8eN_fsu2Gg Addressing the Need for a New Solution In 2024, Nortech announced Expanded Beam Xtreme® as an expansion...
Read More »Wearable medical devices have evolved from simple fitness trackers to sophisticated diagnostic tools that monitor everything from blood...
Read More »High-Performance Flexible Circuit Flex Faraday Xtreme™ Military personnel operate in environments where equipment failure isn’t just inconvenient, it’s...
Read More »